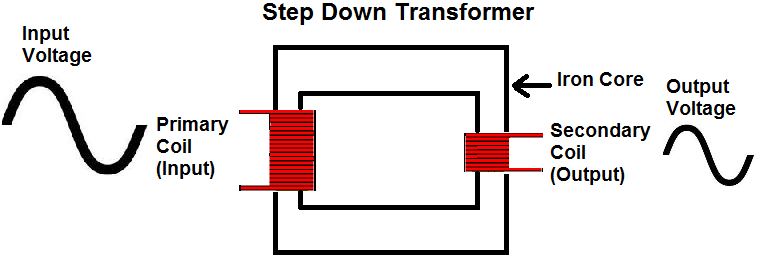
What is a Step Down Transformer?
A transformer is a type of static electrical equipment that transforms electrical energy (from primary side windings) to magnetic energy (in transformer magnetic core) and again to the electrical energy (on the secondary transformer side). A step-down transformer has a wide variety of applications in electrical systems and transmission lines.
A step-down transformer is a type of transformer that converts the high voltage (HV) and low current from the primary side of the transformer to the low voltage (LV) and high current value on the secondary side of the transformer. The reverse of this is known as a step up transformer.
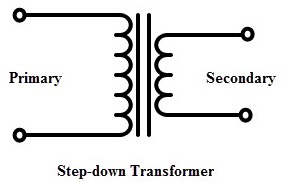
When it comes to the operation voltage, the step-up transformer application can be roughly divided into two groups: LV (voltages up to 1 kV) and HV application (voltages above 1 kV).
Just as transformers can step down the voltage – going from a higher primary side voltage to a lower secondary side voltage – they can also step up the voltage, going from a lower primary side voltage to a higher secondary side voltage. These are known as step-up transformers.
The transformer turns ratio (n) for a step down transformer is approximately proportional to the voltage ratio:

Where VP,S are voltages, and NP,S are the turns numbers on the primary (LV) and secondary (HV) sides respectively. The primary side of a step-down transformer (HV side) has a larger number of turns than the secondary side (LV side).
That means energy flows from the HV to the LV side. The voltage is stepped down from the primary voltage (input voltage) to the secondary voltage (output voltage).
This equation can be rearranged for the formula for the output voltage (i.e. secondary voltage). This is sometimes referred to as the step down transformer formula:

A transformer calculator can help you easily calculate the transformer turns ratio and whether the device is a step down or step up transformer.
The first LV application refers to the transformers in electronic devices. Supplying the electronic circuits requires a low voltage value (e.g. 5V, even lower values nowadays).
A step-down transformer is used to provide this low voltage value which is suitable for electronics supplying. It transforms home voltage (230/120 V) from primary to a low voltage on the secondary side which is used for electronic supplying.
If electronic devices are designed to have higher nominal power, transformers with high operating frequency are used (kHz-s). The transformers with higher nominal power value and 50/60 Hz nominal frequency would be too large and heavy. Also, the daily used battery chargers use the step-down transformer in its design.
Step Down Transformer Applications
The step-down transformers have a very important function in a power system. They lower the voltage level and adapt it to energy consumers. It is performed in several steps described below:
- A long distance energy transmission system should have a voltage level as high as possible. With high voltage and low current, the transmission power loss
will be significantly decreased. A power grid is designed that has to be connected with the transmission system with the different voltage levels. Step-down transformers are used in the interconnection of transmission systems with different voltage levels. They decrease voltage level from high to lower value (e.g. 765/220 kV, 410/220 kV, 220/ 110 kV). These transformers are huge and have very high nominal power (even 1000 MVA). In this case, when the transformer turns ratio is not high the autotransformers are usually installed.
- The next voltage level transformation step is adapting the transmission voltage to the distribution level. The characteristic voltage ratios, in this case, are 220/20 kV, 110/20 kV (also the LV secondary voltages 35 kV and 10 kV can be found). The nominal power of those transformers is up to 60 MVA (usually 20 MVA). The on-load tap changer is almost always installed in these transformers. Voltage regulation is the main function of tap changer. In the USA the tap changer is based on the LV side, and in the rest of the world mostly on the HV transformer side.
- The final voltage transformation step is adapting the voltage to the home voltage level
These transformers are known as small distribution transformers with nominal power up to 5 MVA (mostly below 1 MVA) and with nominal voltage values 35, 20, 10 kV on HV side and 400/200 V on LV side. It is noticeable that those transformers have ahigh turns ratio. They usually have de-energized tap changer with 5 tap position (+/- 2 tap position) and do not have on-load tap changer.


























You must be logged in to post a comment.